Humans have modified food crops for 10,000 years in order to develop varieties with beneficial traits including disease resistance, improved flavor and higher nutrient content. However, the amount of crop improvement that can be achieved through traditional plant breeding is limited because this process can mostly create only new combinations of existing traits.
To create new traits, scientists can induce mutations that change a plant’s genetic code. Mutated genes provide the variety plant breeders need to develop new crops. University of Missouri geneticist Lewis J. Stadler demonstrated in 1927 that X-ray radiation could be used to mutate the DNA of corn and barley. During the 1930s, scientists experimented with this technique to develop new crop breeds.
In the wake of World War II, the desire to use atomic energy for peaceful pursuits in science and medicine grew, and by 1959, scientists in the US, Europe and Soviet Union were irradiating plant seeds in what became known as mutation breeding in hopes of developing improved varieties of wheat, peanuts and many other crops.
Mutagenesis involves exposing plants to ionizing radiation or, more rarely, chemicals to mutate their genes, then growing them for multiple generations to isolate desirable traits. Plants possessing these traits are bred for still more generations to develop uniformity and field tested to see how they perform in real-world settings. If these mutated crops do well, they are licensed and released to farmers.
The International Atomic Energy Agency continues to support mutation breeding research around the world to develop improved crop varieties. More than 2,500 fruits and vegetables consumed today, including barley used to produce Scotch whiskey, disease-resistant cocoa, high yielding rice and spineless pineapples were artificially created with the help of chemical or radiation mutagenesis.
Unlike genetically modified crops which are often created through one or two mutations and are tightly controlled, and although the plant genome is dramatically altered in a laboratory with the induction of thousands of mutations of unknown effect, mutagenized products such as sweet red grapefruit and varieties of durum wheat have not been considered controversial and are often sold as organic. Scientists view mutation breeding as an extension of natural evolution, which is one reason why it has not been tightly regulated and has been embraced by countries around the world.
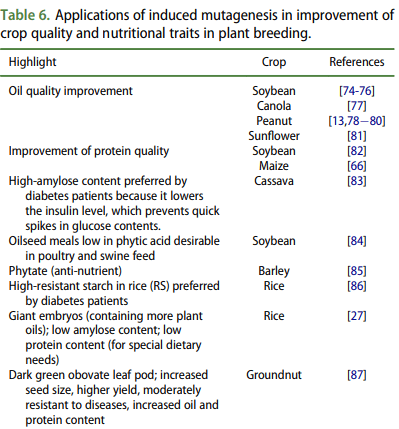
Scientists have often used mutagenesis to improve the nutritional profile of important food crops. The breeding technique has been used to increase the protein content of nuts, corn and soybeans, for example, and to develop diabetic-friendly rice that doesn’t dramatically spike blood sugar. According to a 2016 study, dozens of staple crops have been nutritionally enhanced by mutation breeding.
Some biotechnology critics and organic food advocates who oppose GMO crops have begun taking issue with mutagenesis, claiming it is potentially harmful, and likening it to CRISPR and other forms of gene editing, which they oppose. Many claim CRISPR is yet another secret ploy by “Big Ag” to monopolize the world’s food supply. The popular anti-GMO news site Natural Society warned in 2013:
[The seed company] BASF alone licenses its mutagenesis technology to 40 of the largest seed companies in the world, which turn around and sell mutant seeds to everyone from large ag producers to the farmer down the road. And you, the consumer, who eventually ends up searching for the healthiest option for your family, is none the wiser.
The pro-organic site told its readers that mutagenized crops are more concerning than GMOs because mutagenesis “…. creates random and unspecific genetic changes .” Such criticism is now becoming mainstream in anti-biotechnology circles. The Organic Consumers Association, representing thousands of natural and organic food companies, alleges, “…. mutagenesis can cause dramatic shifts in genetically determined traits, producing unknown toxins or allergens.”
Some advocacy-linked scientists and doctors have speculated about so-called ‘unknown’ or ‘unpredictable’ effects of mutagenesis and gene editing. Cardiologist William Davis, author of the bestselling book Wheat Belly, who claims “modern wheat” is a “perfect, chronic poison,” blames the introduction of dwarf wheat, a crop developed via mutagenesis, for fueling a dramatic increase in gluten allergies.
Such claims get their fuel from a tiny cohort of scientists known for their vocal opposition to biotechnology. King’s College, London molecular geneticist Michael Antoniou, for example, warned in a 2014 report for the anti-biotech group Earth Open Source that mutation breeding during the 20th century “…. could have generated crop varieties that are toxic, allergenic, reduced in nutritional value, vulnerable to pests or environmental stressors, or harmful to the environment.” No evidence supports that claim.
Although mutagenesis does introduce unpredictable changes into a plant’s DNA, this fact isn’t as troubling as some activists maintain. Summarizing the evidence in support of the safety of mutagenesis, the authors of one oft-cited study noted that they were “…. not aware of any indications that the underlying mutations have caused damage to the environment, or had adverse effects on human or animal health.”
Another team of researchers has noted that traditional plant breeding, endorsed by even the most fervent anti-GMO activist, introduces thousands of mutations into the DNA of food crops, yet “[w]e all use and eat such cultivars” without a second thought, they noted.
Philosopher of science Giovanni Tagliabue expanded on these arguments in 2016, explaining there is little significant difference between mutagenesis and other conventional breeding techniques.
[T]he phrase ‘some mutagenized crops have exhibited harmful effects,’ while empirically correct, would not cast any doubt on the technology per se.[W]e should not forget that massive phenotypic consequences – frequently undesirable – can derive from even the slightest change in an organism’s genotype.[S]uch risk of failures from even little tweaks to a genome applies to each and any intervention.”
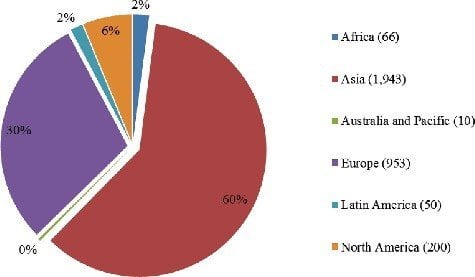
80 years of disaster-free mutation breeding in 30 countries to produce food, animal feed and ornamental plants have convinced food safety officials and scientists around the world that mutagenized crops don’t pose a risk to human health. This is another reason why crops developed through mutagenesis are not regulated in most countries.
Distribution of mutagenized crops by continent:
(Source: Joint Division of the Food and Agriculture Organization of the United Nations and the International Atomic Energy Agency [Joint FAO/IAEA, 2015], [IAEA mutant database, http://mvgs.iaea.org/])
Over the years, other forms of mutagenesis have evolved. Scientists have developed oligonucleotide-directed mutagenesis (ODM), which introduces beneficial changes into a plant’s DNA without generating random mutations caused by traditional chemical and radiation mutagenesis. Plants carrying the desired mutation are then bred as described above and released to farmers.
Site-directed nucleases (SDN) including CRISPR-Cas9, Zinc Finger Nucleases (ZFN) and TALENs are seen by some experts as a refinement of that earlier technology in which the technique can be performed in vivo – in the living organism itself – with much more precision. According to a 2019 study published in Nature:
CRISPR-Cas9 is remarkably specific and efficient at generating on-target genome edits . While CRISPR-Cas9 has the potential to generate off-target cutting in genomic sites that are substantially similar to the target site, off-target edits are likely to be negligible in the background of existing natural variation and continuous unintended changes being generated during the plant breeding process.
These new breeding techniques (NBTs) have added a new layer of complexity to the debate in the European Union (EU) over whether CRISPR is an extension of mutagenesis. While the EU has long recognized the safety of mutagenized crops, it recently determined it will regulate gene-edited and GMO crops as if they were identical. In a July 2018 ruling, the European Court of Justice determined that Europe’s 2001 GMO directive applied to mutagenesis, a reversal of long-standing but unarticulated tradition. It extended that determination to NBTs, but exempted all prior forms of mutagenesis.
[T]he Court of Justice takes the view, first of all, that organisms obtained by mutagenesis are GMOs. [However] the GMO Directive . does not apply to organisms obtained by means of certain mutagenesis techniques, namely those which have conventionally been used in a number of applications and have a long safety record.
The ECJ decision baffled scientists because it created a political distinction between two breeding methods — natural breeding and mutagenesis — whose end results are effectively identical. As biologist Nina Fedoroff explained in the wake of the landmark ruling:
[A]rtificially induced mutations are indistinguishable from naturally occurring mutations at the DNA sequence level. They are also indistinguishable from the genetic changes produced by the newer methods, such as CRISPR/Cas-based gene editing. The indistinguishability of genetic changes made by nature, 20th century mutagenesis and 21st century gene editing highlights the absurdity of regulating genetic modification based on the process used, as the Court of Justice of the European Union ruled in July of 2018.
European plant scientists, whose work was effectively halted by the decision, concurred with Fedoroff’s analysis. The EU’s Group of Chief Scientific Advisors warned that the ruling is likely to block the development of “plants which have the potential to provide immediate direct benefits to the consumer.”
Researchers in the Netherlands delivered a stark confirmation of scientists’ fears in January 2019, announcing they were close to developing a CRISPR-edited, “safe gluten” wheat variety for celiac patients. But, they added, Europe’s prohibitive precautionary-based biotech crop rules would prevent the commercialization of the new variety. Richard Visser, chair of plant breeding at Wageningen University:
Given the current situation in Europe, these wheat genotypes would not be allowed to be grown in Europe.The whole issue surrounding gene editing needs to be settled in Europe.[T]he products made with gene editing techniques should be allowed on the market.
Experts have also warned that excessive, expensive regulation of NBTs, often endorsed by anti-biotech groups, prices universities, nonprofits and small companies that are poised to lead the gene-editing revolution out of the market. This distortion in the marketplace could encourage the very scenario anti-GMO groups claim to fear: A seed market dominated by a few multinational biotech firms.